Product Overview
In Vitro Diagnostic Device for Methotrexate Measurement
Product Description
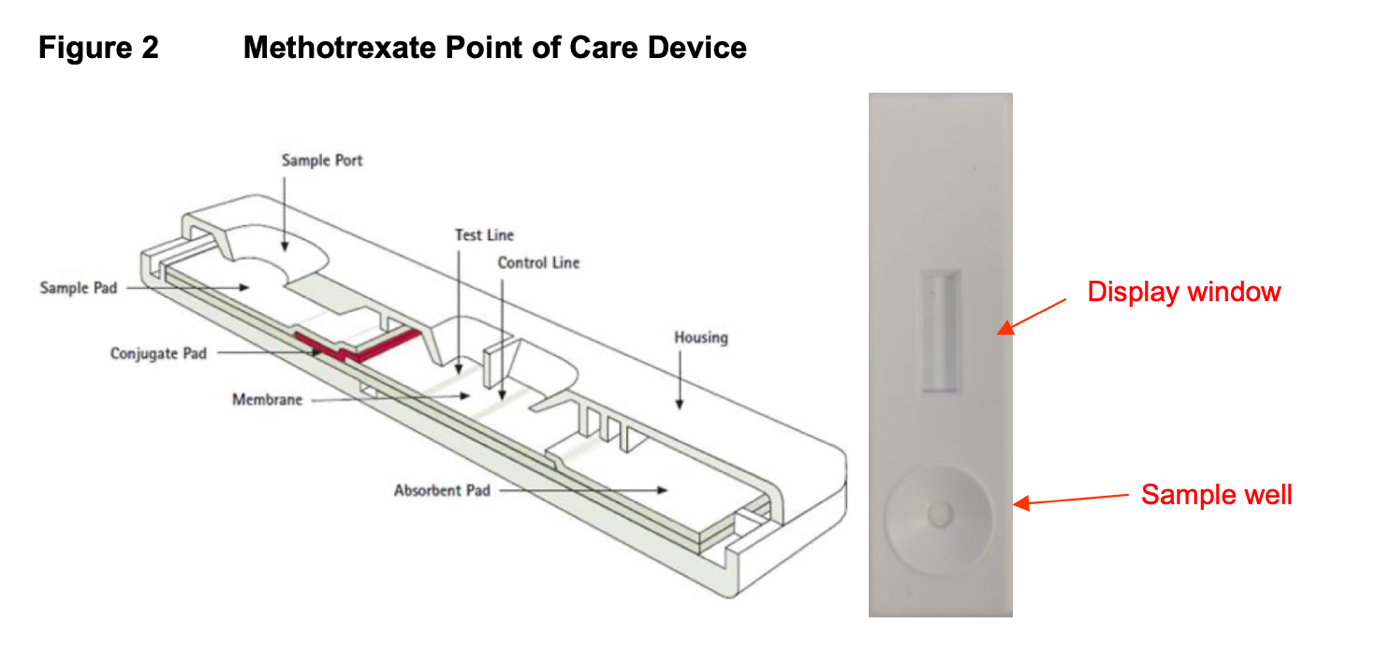
- The product is a lateral flow in vitro diagnostic
- It utilizes a double antibody system in order to achieve a high degree of sensitivity to measuring methotrexate only
- It is able to work in conjunction with off-the-shelf readers to display the result
Technical Specifications
| Indication | Point-of-care diagnostic test intended to detect the levels of Methotrexate (MTX) in whole blood |
| Sensitivity | 0.04 μmol/L |
| Specificity | No cross-reactivity to common blood species, in particular MTX metabolites (e.g. DAMPA) and leucovorin |
| CV % | ≤ 20% CV |
| Detection Range | 0.04–1000 µM in whole blood for detection of MTX |
| Sample Requirements | 40 µl of venous blood (At Launch); capillary blood (Product Life Cycle) |
| Site of Testing | Bedside |
| Time to Result | ≤ 10 minutes |
| Sample Prep & Workflow | Blood collection via lateral flow device → buffer addition |
Intended Use: Resonance MTX Device is a point-of-care diagnostic test intended to detect the levels of Methotrexate (MTX) in whole blood.